GERMINATION.
Not
observed.
MYCORRHIZAE.
Many attempts to establish mycorrhizae of D.
spurca in
one-species pot cultures with Plantago lanceolata L. as the plant
host failed. According
to Morton (2000) and Pfeiffer et al. (1996), the mycorrhizae of D. spurca
(considered as Glomus spurcum C.M. Pfeiff., C. Walker & Bloss emend. L.J. Kenn., J.C. Stutz & J.B. Morton) consisted of arbuscules and intraradical hyphae staining with variable intensity
(from almost invisible to dark). No vesicles were found.
DISTRIBUTION.
In
Poland, spores of D. spurca were first found in three trap cultures
with root-rhizosphere soils of Beta vulgaris L. and Triticum
aestivum L. cultivated in Drzemin (53º15’N, 14º39’E),
Kolbacz (53o18’N, 14o49’E), and Stobno (53º26’N,
14º23’E) in the Western Pomerania voivodeship (Blaszkowski et
al. 2003; Iwaniuk and Blaszkowski, unpubl.). Subsequently, this fungus was
revealed in 40 trap cultures containing root-rhizosphere mixtures taken
from under Corynephorus canescens (L.) P. Beuv., Elymus arenarius
L., Eryngium maritimum L., Festuca rubra L., Phragmites
australis (Cav.) Trin. ex Steud., Rubus fructicosus L. nom.
Ambig., and Salix sp. colonizing maritime dunes adjacent to Darlówko
(54º26’N, 16º23’E) adjacent to the Baltic Sea.
Diversispora spurca has originally been discovered as Gl. spurcum in a greenhouse bed of sand
used for propagation of various ornamental plants cultivated in Arizona
(Pfeiffer et al. 1996). This fungus has also been found in maritime dunes
of Mexico (Pfeiffer et al. 1996), Hawaii (Koske and Gemma 1996), San Miguel
Island, California (Koske, pers. inform.), as well as in different other
natural ecosystems of North America, Cuba and Namibia, Africa (Kennedy et
al. 1999; Stutz and Morton 1996; Stutz et al. 2000).
NOTES.
The new combination D. spurca has been erected from Gl. spurcum mainly based on the molecular separateness of this fungus compared with other members of the genus Glomus (Walker and Schüßler 2004).
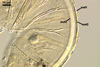
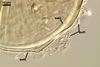
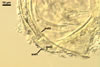
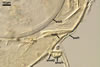
The layer 1 of spores of D. spurca
characterized here has been not included in the spore wall structure of
this fungus in any earlier its description (Blaszkowski et al. 2003; Kennedy
et al. 1999; Pfeiffer et al. 1996), although the presence of this structure
has been mentioned. Pfeiffer et al. (1996) considered it to be a mucilaginous
material formed by overlapping plate-like structures when examined with
a scanning electron microscope. According to Kennedy et al. (1999), it rather
is an accumulation of organic matter, because it does not stain in Melzer's
reagent as a typical mucilaginous layer.
The
wall layer 1 of D. spurca spores examined by the author of this
website occurred in almost all the specimens of this fungus coming from
both Poland and those received from Dr. C. Walker, U. K.
The fungal species
most similar morphologically to D. spurca are Gl. albidum
C. Walker & L.H. Rhodes, Gl. eburneum L.J. Kenn. et al., Gl.
gibbosum Blaszk., Gl. viscosum Nicol., and Paraglomus
occultum (C. Walker) J.B. Morton & D. Redecker. These species produce
hyaline to light-coloured spores of a similar size when observed under a
dissecting microscope.
However, Gl. albidum
forms spores with a wall composed of an outer evanescent layer and an inner
laminate layer (Walker and Rhodes 1981). Both layers have a similar thickness.
In contrast, the outer layer of D. spurca spores is permanent
and much thinner than the inner laminate layer. Young spores of Gl.
albidum stain pink to orange in Melzer’s reagent, whereas those
of D. spurca are non-reactive in this reagent.
The property distinguishing
Gl. eburneum is the formation of ovoid to tear-drop shaped spores
(Kennedy et al. 1999). Spores of D. spurca consistently are globose.
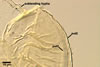
However, the main differences between these fungi reside in the properties
of spore wall layers, which are evident only in heavily crushed spores.
The outer layer of Gl. eburneum spores remains adherent to the
inner laminate one, and the permanent layer 1 and the laminate layer 2 of
the D. spurca spore wall usually completely separate from each
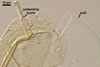
other. Additionally, most vigorously crushed spores of D. spurca
are devoid of subtending hypha that along with layer 1 of spore wall separates
from the laminate spore wall layer forming too weak structural support of
the hypha.
Layers 2 and 3 of
spore wall of Gl. gibbosum are similar to layers 1 and 2 of spore
wall of D. spurca (Blaszkowski 1997). However, in the former
species, layer 2 is still associated with an outer evanescent layer, and
the laminate layer 3 surrounds a flexible, colourless innermost layer that
is lacking in the latter fungus.
Spores of Gl.
viscosum occur in loose aggregates (Walker et al. 1995) rather than
singly in the soil as those of D. spurca. The spore wall of Gl.
viscosum consists of three layers (Morton 2000) with the outermost
one exuding a mucigel-like substance (this phenomenon does not occur in
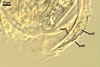
D. spurca). Glomus viscosum also has a persistent subtending
hypha (vs. it usually is absent in crushed spores of D. spurca).
Spores of P. occultum
remain hyaline throughout their life cycle (Morton and Redecker 2001; Walker
1982). In contrast, mature spores of D. spurca usually are pale
yellow. Additionally, the wall of P. occultum spores consists
of three layers of equal thickness, whereas the total thickness of the wall
layers 1 and 2 of D. spurca spores is much lower than the thickness
of the innermost layer 3. Finally, the innermost layer of P. occultum
is a permanent structure lacking sublayers (vs. a laminate layer in D.
spurca).
REFERENCES
Blaszkowski J. 1997.
Glomus gibbosum, a new species from Poland. Mycologia 89, 339-345.
Blaszkowski J., Adamska
I., Czerniawska B. 2003. Glomus claroideum and G. spurcum,
arbuscular mycorrhizal fungi (Glomeromycota) new for Poland and Europe,
respectively. Acta Soc. Bot. Pol. 72, 149-156.
Kennedy L. J., Stutz
J. C., Morton J. B. 1999. Glomus eburneum and G. luteum,
two new species of arbuscular mycorrhizal fungi, with emendation of G.
spurcum. Mycologia 91, 1083-1093.
Koske R. E., Gemma
J. N. 1996. Arbuscular mycorrhizal fungi in Hawaiian sand dunes: Island
of Kaua’i. Pacific Sci. 50, 36-45.
Morton J. B. 2000.
International Culture Collection of Arbuscular and Vesicular-Arbuscular
Mycorrhizal Fungi. West Virginia University.
Morton J. B., Redecker
D. 2001. Two families of Glomales, Archaeosporaceae and Paraglomaceae, with
two new genera Archaeospora and Paraglomus, based on concordant
molecular and morphological characters. Mycologia 93, 181-195.
Pfeiffer C. M., Walker
C., Bloss H. E. 1996. Glomus spurcum: a new endomycorrhizal fungus
from Arizona. Mycotaxon 59, 373-382.
Stutz J. C., Morton
J. B. 1996. Successive pot cultures reveal high species richness of arbuscular
mycorrhizal fungi in arid ecosystems. Can. J. Bot. 74, 1883-1889.
Stutz J. C., Copeman
R., Martin C. A., Morton J. B. 2000. Patterns of species composition and
distribution of arbuscular mycorrhizal fungi in arid regions of southwestern
North America and Namibia, Africa. Can. J. Bot. 78, 237-245.
Walker C. 1982. Species
in the Endogonaceae: a new species (Glomus occultum) and a new
combination (Glomus geosporum). Mycotaxon 15, 49-61.
Walker C., Rhodes L.
H. 1981. Glomus albidus: a new species in the Endogonaceae. Mycotaxon
12, 509-514.
Walker C., Giovannetti
M., Avio L., Citernesi A. S., Nicolson T. H. 1995. A new fungal species
forming arbuscular mycorrhizas: Glomus viscosum. Mycol. Res. 99,
1500-1506.
Walker C., Schüßler A. 2004. Nomenclatural clarifications and new taxa in the Glomeromycota. Mycol. Res. 108, 979-982.